Navigating the spectral slopes of the NSRR cohorts
Nataliia Kozhemiako & Shaun Purcell
We recently used NSRR data to study the so-called spectral slope of the electroencephalogram (EEG), during wake, NREM and REM sleep. This work (described in a bioRxiv pre-print, Kozhemiako et al.) points to the power of NSRR data: thousands of polysomnographic studies that can be rapidly and freely repurposed to address a broad array of research questions. Nonetheless, there are challenges inherent in any analysis of existing data, meaning that (surprise, surprise...) careful analytic approaches are of prime importance. In this blog, we briefly reflect on both sides of this coin: the opportunities afforded by NSRR data, coupled with a few of the obstacles that we had to navigate around.
Specifically, below we:
- outline the scientific question and our results
- underscore some general issues involving choice of mastoid reference and high frequency EEG
- document a few technical issues in the Sleep Heart Health Study that we discovered in the course of this project
Here we repeat some of the practical and technical issues mentioned in Kozhemiako et al., to highlight these issues for users of NSRR data.
Brain activity and the spectral slope
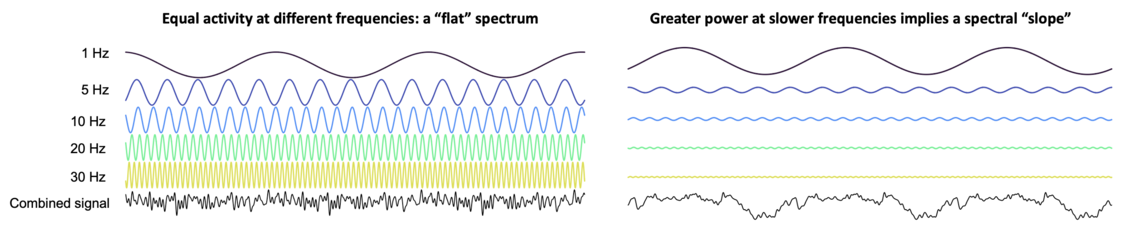
As given by the Fourier transform, the power spectrum of the human EEG has a characteristic slope, reflecting the tendency for activity at slower frequencies to be of higher amplitudes, and vice versa. In the cartoon below (which is not a realistic depiction of EEG data on any level), the left panel shows a scenario in which this is not the case. Here, activity levels have similar amplitudes (heights) across all frequencies (e.g. 1 Hz, 5 Hz, etc). The lower black signal depicts the summation of these components, i.e. the hypothetical observed EEG. In this scenario, the associated power spectrum (plotting frequency on the x-axis and power/activity on the y-axis) would be a relatively flat, horizontal line, indicating a similar degree of activity across all frequencies.
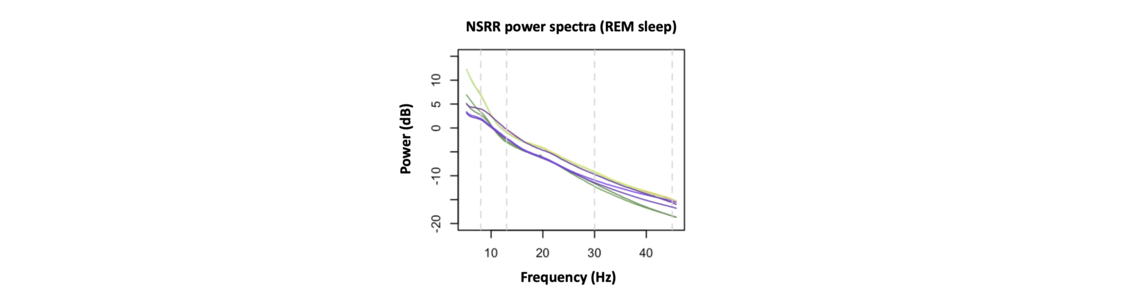
In contrast, the right panel reflects a different scenario, in which activity at faster frequencies tends to have reduced amplitude. As it turns out, this loosely mirrors what we actually see in real data. The corresponding power spectrum will have a downward slope (going left-to-right from slower to faster frequencies). The figure below shows EEG power spectra from several NSRR cohorts during REM sleep (each line is the average spectrum for that cohort), and all clearly exhibit the characteristic downward slope.
The mathematical and conceptual underpinnings of the spectral slope are beyond the scope of this post: suffice to say, there is reason to believe that the steepness of the spectral slope reflects different modes of brain functioning. Previous studies have shown that states of reduced arousal level (e.g. being asleep or under general anesthesia) are associated with steeper slopes. Most relevant to the current work, Lendner et al. (2021) recently reported that REM sleep has an even steeper slope compared to both NREM sleep and wake. This is interesting because REM sleep is typically considered to be "wake-like" in terms of its associated brain activity (which is why it is sometimes called paradoxical sleep).
As the original publication was based on a small sample size, we wanted to use openly available NSRR studies to replicate and potentially extend some of Lendner et al.'s observations. Using the Luna toolset to perform the analyses, this work is described in a bioRxiv pre-print Kozhemiako et al. (2021). As well as successfully replicating the original finding (in a sample several orders-of-magnitude larger), we were able to describe some other aspects of the sleep EEG that, along a person's average spectral slope, exhibited qualitatively different patterns between wake and REM.
As described in Kozhemiako et al. (2021), however, our initial analysis of NSRR datasets (CHAT, CCSHS, CFS, SHHS, MrOS and SOF) went through a few iterations, with adjustments made to account for some initially unappreciated aspects of these data. As noted, two issues that we feature here are 1) the general impact of mastoid reference choice, and 2) technical issues specific to the SHHS that impact the high frequency (e.g. >20 Hz) EEG.
Mastoid referencing and muscle/cardiac contamination in the EEG
As detailed in Kozhemiako et al. (2021) (in particular, in the Supplementary Information), we originally based analyses on central EEG channels using contralateral mastoid referencing: e.g. the left central C3 electrode referenced to the right mastoid, M2. Contralateral mastoid referencing is common for polysomnographic data and is recommended by the American Academy of Sleep Medicine (AASM) for scoring clinical studies. We nonetheless paid close attention to the referencing scheme used, because 1) our analysis focused on frequencies above 30 Hz, at which the EEG is known to be more susceptible to muscle and/or cardiac contamination, and 2) mastoid electrodes are known to be particularly susceptible to these classes of artifact.
We conducted a round of secondary analyses looking at the slopes of the EMG and ECG as well as the EEG, and also the coherence between EMG/ECG and EEG signals (indexing possible contamination of the EEG by non-neural sources). Indeed, our results suggested higher levels of artifact when using contralateral mastoids, compared to the alternative of bilateral linked mastoid referencing: e.g. C3 referenced against the average of M1 and M2. Likewise, using a bipolar channel (e.g. C3 referenced against C4) also appeared to ameliorate these issues.
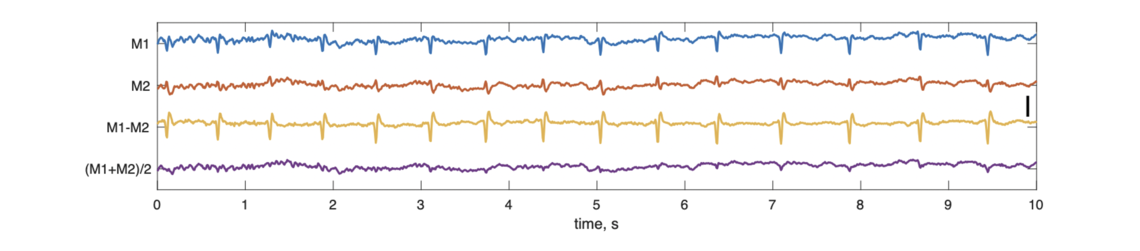
With respect to cardiac interference at mastoid electrodes, the figures below show a case where linked mastoid referencing clearly reduces artifact. This first figure shows 10 seconds of signal from the mastoids as originally recorded (i.e. referenced against the Fpz electrode).
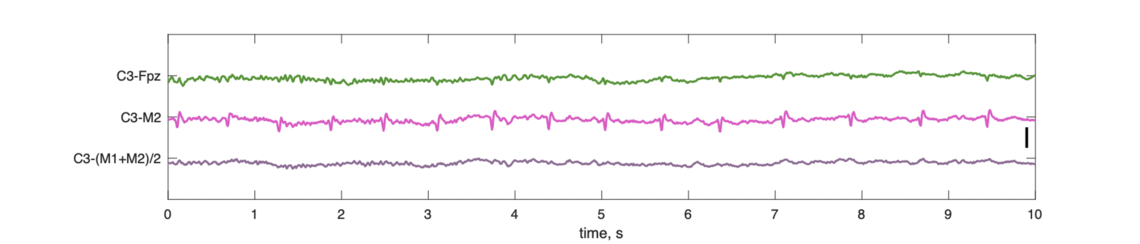
Both M1 and M2 are clearly impacted by cardiac activity (spikes evident in the traces). The average of the mastoids (lower trace) almost fully eliminates this issue, as the artifacts in M1 and M2 effectively cancel each other out. (In contrast, the M1-M2 "cross-mastoid", included in the manuscript for QC purposes, exacerbates it.). Consequently, the central channel (C3) referenced against the linked mastoid (lower trace) avoids this artifact being introduced, whereas this is not the case for the single contralateral mastoid (middle trace):
Naturally, issues of artifact in the EEG, and the role of referencing to ameliorate them, are well known. However, as we previously documented in our work on sleep spindles in NSRR cohorts, even subtle artifacts not evident on visual inspection can bias results. Just as large samples afford greater statistical power to detect true effects, they inevitably bring greater risk of detecting spurious effects too, which can be problematic if such effects are related to demographic or clinical factors, including age, sex or body mass index.
In the spectral slope manuscript, we show that although statistically similar, results based on linked mastoid referencing appeared to be more robust to these types of artifacts than those based on contralateral mastoid referencing. If the spectral slope or other EEG metrics are ultimately to be used as clinically interpretable biomarkers, understanding these sources of variation (whether due to bias, noise or true signal) will be important.
The bottom line: especially if you are performing analyses that include high-frequency (e.g. >30 Hz) EEG information, you may want to pay particular attention to the choice of referencing scheme. However, bear in mind that there is no one-size-fits-all correct approach, and different analyses and/or different datasets may benefit from different choices.
Technical issues in the Sleep Heart Health Study data
The SHHS data were collected over 30 years ago, using first-generation portable devices that could be more prone to artifact than typical devices on the market today. We discovered a set of technical issues that were specific to both SHHS datasets and had a marked impact on our analyses of spectral slopes, but could conceivably impact other types of analyses, particularly those that (directly or indirectly) use higher frequency EEG information.
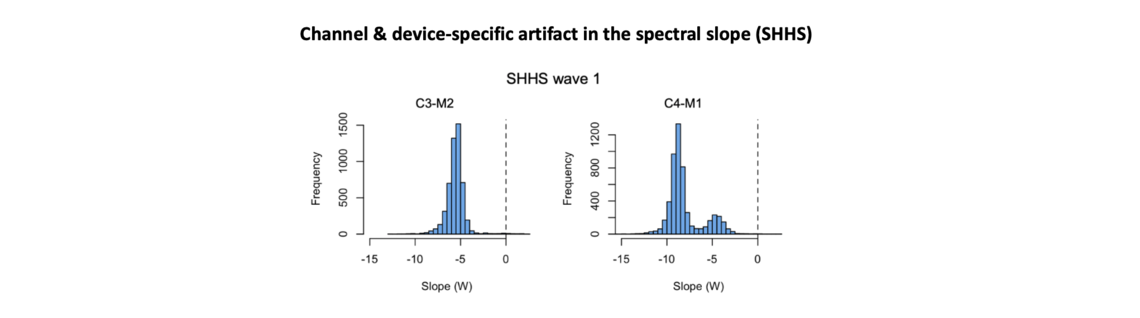
We found that all SHHS studies had much steeper spectral slopes than expected: the SHHS was a clear outlier compared to all other NSRR cohorts in this regard. On top of this, we observed two further indications of technical problems. First, C4-M1 slopes were, on average, considerably steeper than C3-M2 slopes (in contrast, all other cohorts had near identical slope distributions for the two central channels). Second, for C4-M1 but not C3-M2, there was a clear bimodality in the slope distribution, seen in both SHHS1 and SHHS2 datasets (in contrast, all other cohorts showed broadly normal slope distributions):
The C4-M1 slope bimodality was not accounted for by any obvious demographic, person-centric factor. On investigation, the physical recording device ID appeared to account for this effect, instead. The initial SHHS1 study employed around 50 physical units: of these, 6 or 7 had markedly different average slope estimates for C4-M1, compared to the other units. (Note that, compared to other NSRR cohorts, all SHHS devices produced outlying results however, this issue of device-related bimodality notwithstanding.)
Other than a 0.15 Hz high-pass filter, no digital filtering was subsequently applied to the SHHS EEG that could straightforwardly account for these differences. For whatever set of technical reasons, the Compumedics P-series units used in this study appeared to have very non-flat frequency response curves from 0.5 to 50 Hz, which also varied between channels and units, and biased estimates of spectral slopes. For our analyses of the 30-45 Hz spectral slope, we therefore excluded both SHHS datasets.
(Note that the SHHS devices had hard-wired contralateral mastoid references, i.e. C4-M1 and C3-M2 being labelled EEG and EEG2 respectively: we could not explore whether alternative mastoid or bipolar referencing might have mitigated these issues.)
The bottom line: if you are using SHHS data for analyses that leverage the "high" (in this context, say >20 Hz) frequency EEG, you should be aware of this set of issues. See the Supplementary Informaton of Kozhemiako et al. (2021) for more details. We plan to make a list of the device/headbox IDs for each SHHS1 and SHHS2 participant available soon.